Fayetteville startup awarded patent for healthcare device
by June 9, 2020 2:13 pm 1,805 views
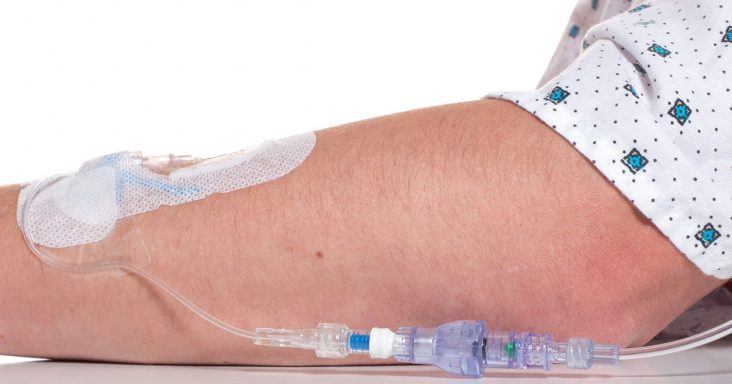
A SafeBreak Vascular installed in an IV line.
Fayetteville-based healthcare technology company Lineus Medical has received a U.S. utility patent for key features of its breakaway medical device technology that helps protect IV lines.
The patent is for the company’s flagship product, SafeBreak Vascular. This is the third patent awarded to Lineus Medical for the device, which has two utility patents and one design patent.
SafeBreak Vascular is the first breakaway medical device designed for peripheral IV lines, according to a news release. When force is applied to an IV line, SafeBreak Vascular separates to reduce the effects of the force and prevents patient bleeding and medication spillage.
“This issuance of this new utility patent is another important step in adding value to our company and protecting our first product to market, SafeBreak Vascular,” CEO Vance Clement said. “The portfolio of patents the company has secured puts the company in a strong intellectual property position to protect and defend its value and SafeBreak Vascular from competition.”
A hospital patient receives an IV line between 60% and 90% of the time, and peripheral IV lines have a 46% fail rate, according to the Lineus Medical website. When IVs are dislodged, the failures are a result of excessive force being applied to the IV line.
“We continue to build an intellectual property moat around SafeBreak Vascular, and that moat just became more difficult to overcome,” said Spencer Jones, chief technology officer and founder of Lineus Medical. “Innovation in product design must be accompanied by meaningful patent protection, and we expect that our issued patents currently under review will further protect our technologies and set us apart for many years in the market place.”
SafeBreak Vascular is not yet for sale and does not have clearance from the U.S. Food and Drug Administration.
